 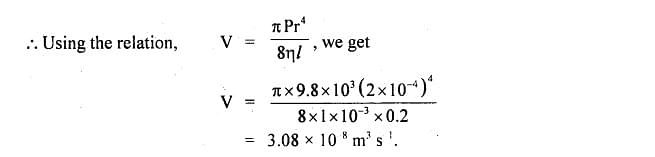
All four parameters listed above have to be calculated as a function of critical properties in order to apply Equation (19.26). Here it is written as originally proposed by Jossi, Stiel and Thodos (1962). original paper presents a typographical error in Equation (19.26). See Technical Requirements in the Orientation for a list of compatible browsers. Ρ r This equation is not rendering properly due to an incompatible browser. 
Ξ m This equation is not rendering properly due to an incompatible browser. = viscosity at atmospheric pressure (c p), 
Μ ∗ This equation is not rendering properly due to an incompatible browser. Μ This equation is not rendering properly due to an incompatible browser. Such expression, originally proposed by Jossi, Stiel and Thodos (1962) for the prediction of the viscosity of dense-gas mixtures, is given below: Lohrenz, Bray and Clark (1964) proposed an empirical correlation for the prediction of the viscosity of a liquid hydrocarbon mixture from its composition. This serves the purpose of guaranteeing that the gas phase and condensate phase converge to the same value of viscosity as they approach near-critical conditions. It this type of simulation, it is usual to calculate gas viscosities based on Lohrenz, Bray & Clark correlation as well. However, in compositional simulation, where both gas and condensate compositions are known at every point of the reservoir, it is customary to calculate condensate viscosity using Lohrenz, Bray & Clark correlation. Corrections must be applied for under-saturated systems and for systems where dissolved gas is present in the oil. The most commonly used oil viscosity correlations are those of Beggs-Robinson and Vasquez-Beggs. ) in lbm/ft 3 (calculated at the pressure and temperature of the system), and the resulting viscosity is expressed in centipoises (c p). In this expression, temperature is given in (°R), the density of the fluid ( ρ g This equation is not rendering properly due to an incompatible browser. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
